By BIOSYNTH AG
BIOSYNTH Molecule of the Month: Amygdalin
Introduction
Microbiology and organic fine biochemical CDMO and specialist supplier, BIOSYNTH AG, has more than 100, 000 substances and molecular entities in its product portfolio. Each of them has its own particular applications and story to tell.
Here is an example of the molecular entities that BIOSYNTH regularly features on its website every week. This month we are focusing on amygdalin – the substance that gives cyanide its notorious ‘bitter almonds’ signature but also a source of essential trace nutrients.
Amygdalin: description
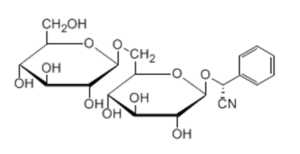
Amygdalin (Biosynth catalog number A-7800) is also known as D Mandelonitrile-beta-gentiobioside or D-Mandelonitrile-beta-D-glucosido-6-beta-D-glucoside, with the chemistry formula C20H27NO11. It is a cyanogenic glycoside derived from the aromatic amino acid phenylalanine.
Amygdalin is found in many plants, but most notably in the seeds (kernels) of apricot, bitter almonds, apple, peach, and plum. Within these plants, amygdalin and the enzymes necessary to hydrolyze them are stored in separate locations so that they will mix in response to tissue damage. This provides a natural defense system.
Amygdalin is classified as a cyanogenic glycoside because each amygdalin molecule includes a nitrile group that can be released as the toxic cyanide anion by the action of a beta-glucosidase: eating amygdalin will cause it to release cyanide in the human body, and may lead to cyanide poisoning.
Amygdalin in history
Amygdalin was first isolated as a compound from bitter almonds during the 1830s, initially considered non-toxic.
From the early 1950s, amygdalin was notoriously promoted as an alternative cancer treatment, under the misnomer vitamin B17, first in Russia and a few years later in the US in a semi-synthetic, injectable form, patented as Laetrile.
It became the most commonly used non-conventional anti-cancer treatment but soon, studies found both amygdalin and laetrile to be clinically ineffective, not to mention potentially toxic. Researchers found that amygdalin taken orally undergoes enzymatic hydrolysis via β-Glucosidase and is converted to two glucose molecules as well as mandelonitrile, which due to its poor stability is spontaneously converted to hydrogen cyanide and benzaldehyde, opening up risk of . cyanide poisoning. The promotion of laetrile as an anti-carcinogenic has been described in medical literature as “the slickest, most sophisticated, and certainly the most remunerative cancer quack promotion in medical history”.
Amygdalin in modern medicine
However, this does not mean amygdalin has no therapeutic value, even though it is most emphatically not a vitamin.
The fruit kernels that contain amygdalin are known to have a wide range of positive effects on human health.
In nature, stone fruit kernels are actually important sources of proteins, carbohydrates, minerals, vitamins, phenolics and liphophilic bioactive compounds, such as phytosterols, tocopherols, carotenoids, sterols and squalenes.
In fact, the high nutritional value of the fruit kernels makes them highly attractive to herbivores. Thus plant synthesis of amygdalin can be seen as a natural defense mechanism.
Resources
Click on BIOSYNTH Molecule of the Month: Amygdalin for more information.
Click on BIOSYNTH to contact the company directly.
Click on Amygdalin: A-7800 for product information.
Supplier Information
Supplier: BIOSYNTH AG
Address: Rietlistr. 4, 9422 Staad, Switzerland
Tel: +41 (0)71 858 20 20
Fax: +41 (0)71 858 20 30
Website: www.biosynth.com