By Progress Project…
Progress-PME Case Study: Fast track project: from feasibility study to completed qualification and plant handover
Progress Project Management & Engineering (Progress-PME) provided project management expertise and specialized knowledge of risk management and fast track principles to help a Netherlands-based pharma client create a new cGMP production facility in just over 18 months.
Client needs
Tiofarma, based in the South Holland town of Oud-Beijerland, is a pharmaceutical production company that manufactures generic medicines such as tablets, capsules, creams, ointments and drinks, on a small and medium scale for customers across the EU.
Faced with growing demand and ever-more stringent regulatory requirements (GMP/FDA), Tiofarma launched in January 2015 the project to convert one of its former warehouses into an ultra-modern and GMP compliant production facility for creams and ointments.
Collaborative strategy
Over the following 20 months, Progress-PME executed all aspects of conceptual engineering and project management on construction of the new facility. After completing the conceptual design for this multi-product plant, Progress-PME helped the client create and follow a team-based approach to construction, forming an active and dynamic collaboration between client, building contractor and advisors.
The construction team strategy is based on proactive cooperation between the different parties and an integrated approach ensuring optimum coordination between the various disciplines.
This strategy reduces cost, lead time and increases overall quality. Progress-PME also advised Tiofarma in the selection and recruitment of construction team partners.
Project management
Progress-PME emphasized a joint team approach and shared ownership of the new facility during the project. The Tiofarma project team comprised a multi-disciplinary mix of Tiofarma employees and external specialists.
While construction and main design of the revised building and principle installations were executed by the main contractor, other members of the project team were responsible for specification, sourcing, and ordering of key components and process equipment, all handled as ‘just in time’ deliveries to main contractor for installation.
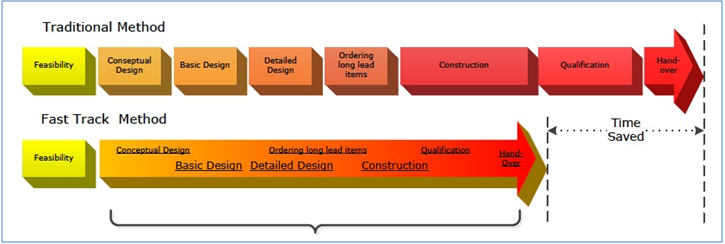
Fast track principles
The facility was built, commissioned and qualified according to fast-track principles. This meant that, rather than use consecutive sequencing, progress was accelerated by executing various activities in parallel, using risk management principles.
Principles from project management by PRINCE2 (Projects In Controlled Environments) and Lean Six Sigma were applied to ensure that the design, construction and qualification of the equipment was realized in the shortest possible time within the targets for quality and budget.
These methodologies were applied to ensure that the design, construction and qualification of the equipment was realized in the shortest possible time within the quality and budget targets.
This structured approach, in close collaboration of Tiofarma employees and external contractors, combined with a high degree of involvement of the project team to make this project a great success. Following an inspection by the Netherlands’ IGZ Healthcare Inspectorate, the manufacturing licence for the new facility was issued in August 2016, even while the last validation activities were being completed.
Qualification & validation
Qualification and validation was executed following the most recent guidelines from EMA and FDA. Progress also delivered the validation manager and validation engineers to work together with installation companies and Tiofarma employees during commissioning and validation phases.
This approach succeeded in effecting a smooth transfer of the new facility and its installations from external companies to internal employees.
The client commented: “The project management was set up and implemented very professionally. What’s more, the project team managed to find the perfect balance in terms of timing, costs and quality and was responsible for the close involvement of all the employees in the Tiofarma departments.”
About Progress-PME
Progress-PME is an independent Project Management and Consultancy Company with customers in life science, (bio) pharmaceutical, medical device and healthcare industries. The company was founded in 1999, operates internationally and provides support throughout the entire range of process development to commercial production.
Progress-PME provide services in the field of project and interim management and Consultancy. Fields of expertise: CMC, Quality (cGxP), Validation, Engineering and LEAN Six Sigma.
Resources
Click on Fast track project to download fact sheet.
Click on Lean Six Sigma for further information.