By Novo Nordisk…
Novo Nordisk Pharmatech Vaccine Support
Innovative Danish manufacturer Novo Nordisk Pharmatech is a renowned supplier of very high quality CTAB (Cetrimonium Bromide), which is a powerful cationic surfactant that can be used in vaccine downstream processing in both bacterial and viral vaccines.
Furthermore, Novo Nordisk Pharmatech recombinant insulin promotes cell growth and viability in manufacturing of cell-based vaccines.
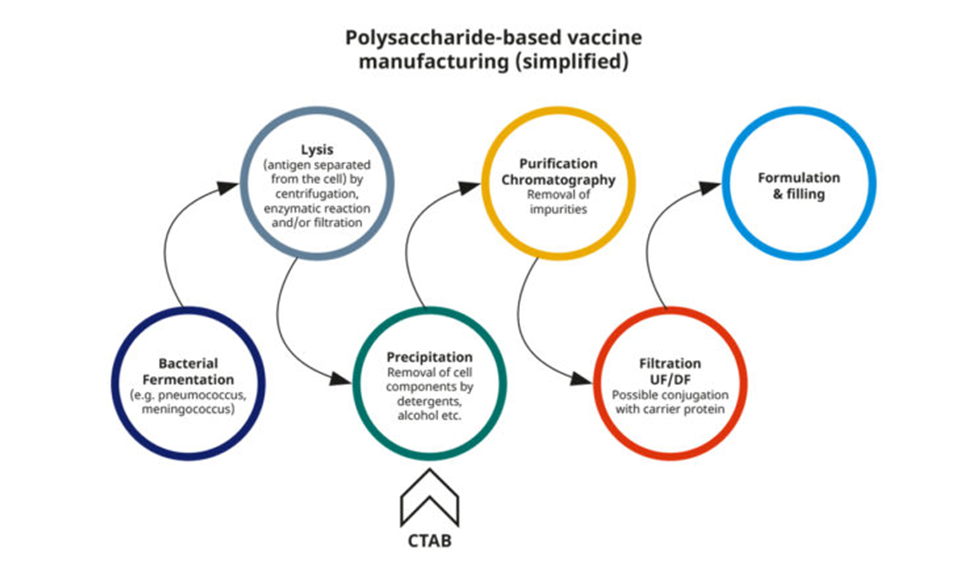
Role of CTAB in bacterial, polysaccharide-based vaccines
The polysaccharide vaccines commonly found in standard children vaccination programs are inactivated subunit vaccines composed of long chains of sugar molecules, that make up the surface capsule of certain bacteria. These vaccines typically target pneumococcal and meningococcal diseases.
The WHO recommends use of CTAB in bacterial vaccine manufacturing as a purification agent for polysaccharide vaccines, where it serves as a surfactant in a buffer system, to remove membrane lipids and promote cell lysis, to “free-up” the polysaccharides.
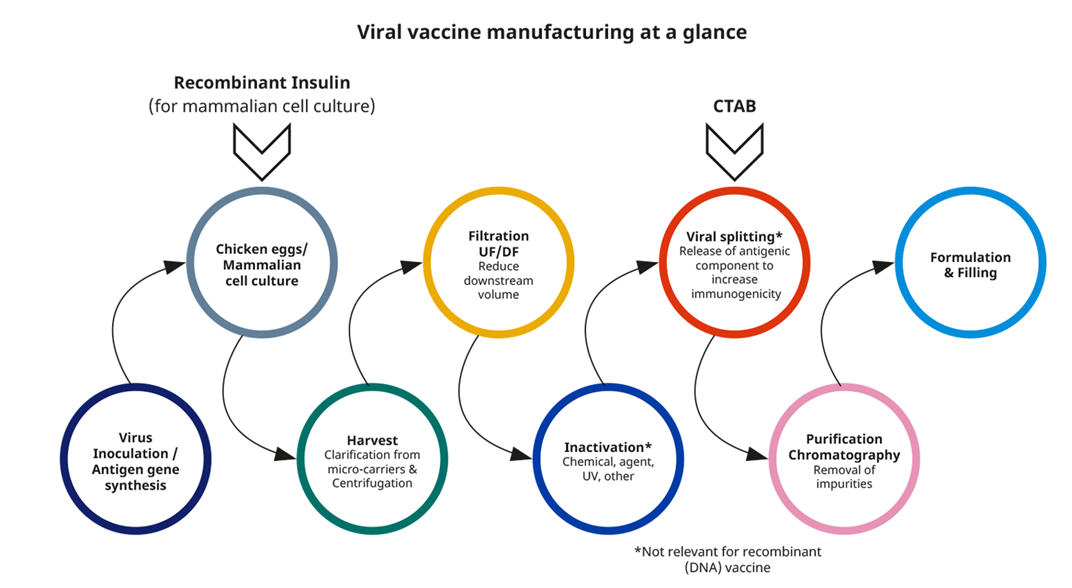
Role of CTAB and Recombinant Insulin in viral vaccine manufacturing
Novo Nordisk Pharmatech’s Insulin Human AF and CTAB can also be used in viral vaccine manufacturing, such as influenza vaccines.
lnsulin Human AF, also known as recombinant insulin, consists of human insulin crystals, a biosynthetic product produced by recombinant microbial expression in yeast. It is a key upstream component in serum free growth media for mammalian cells, stimulating cell proliferation and enhancing the yield of the cell cultures used for viral vaccines. Insulin Human AF is, approved by regulatory bodies worldwide, including FDA and EMA. It is also used in manufacturing of monoclonal antibodies (mAbs), gene therapy products and other biological drug products.
Further downstream, CTAB is used as a cationic detergent to isolate antigenic agents, such as influenza HA proteins and for purification of egg-based, cell-based and recombinant (DNA) vaccines. Sub unit isolation can be done using a mixture of Tween 80 with CTAB, to separate RNP (Ribonucleoprotein) particles from the surface antigen proteins by centrifugation.
CTAB benefits
Novo Nordisk Pharmatech is the only quaternary ammonium compound (Quat) manufacturer offering CTAB in cGMP manufactured pharmaceutical grade, in full compliance with ICH Q7.
Tangible user benefits include:
- Full traceability
- High product purity
- Full USP/NF compliance
- Audit access
- Full regulatory documentation package
- Manufactured under fully validated processes
Insulin Human AF benefits
Novo Nordisk Pharmatech Insulin Human AF is manufactured in cGMP facilities by Novo Nordisk A/S in accordance with its own quality system, based on ISO 9001 and cGMP. The product is analyzed according to the current European Pharmacopoeia (Ph. Eur.) and United States Pharmacopoeia (USP).
Product advantages include:
- Pure and animal-free recombinant insulin
- Consistent high quality
- Full traceability
- Secure global supply chain
- Extensive regulatory documentation
- Continuous availability
- High levels of service and support
- Cost-effective customized solutions
- FDA-inspected manufacturing facilities
- Safety stock at multiple secured locations
- Large-scale bulk batches solely for the cell growth market
- Five-year shelf life
- Robust risk mitigation strategy
Resources
Click on Quats applications in vaccine production to learn more.
Click on Human Insulin as cell growth medium to learn more.
Click on Novo Nordisk Pharmatech pharmaceutical grade quaternary ammonium compounds to download product brochure.