By Novo Nordisk…
Novo Nordisk Pharmatech showing recombinant insulin advances at BPI West in California
Koege, Denmark: – Novo Nordisk Pharmatech A/S, the leading worldwide supplier of high-quality insulin, will be a leading presence at the upcoming BioProcess International (BPI) US West Coast (BPI West) meeting in Santa Clara, California from March 11th – 14th.
Novo Nordisk Pharmatech’s appearance at BPI West will come hard on the heels of its presence on the other side of the Pacific at Biologics Manufacturing Asia in Singapore.
At both events, Novo Nordisk Pharmatech will focus on latest developments in its powerful recombinant insulin human animal-free (AF) platform for cell culture, particularly its potential to increase viable Chinese hamster ovary (CHO) cell density and to revolutionize the supply chain for CAR-T therapies.
Increased CHO cell density
“CHO cells are one of the most widely used platforms for the production of biopharmaceuticals. Increased demand for safety and reliability has moved the standard for CHO cell culture media from Serum to Serum free and further on to chemically defined media,” commented Novo Nordisk Pharmatech. Product Manager, Sara Gualdoni, who will be present in both Singapore and Santa Clara.
“As the world’s largest supplier of recombinant insulin, we have shown In collaboration with UAB that adding animal origin-free insulin to three leading commercially available off-the-shelf chemically defined media have shown significant increases in viable cell density. In addition, insulin has been proven to aid in the expression of difficult-to-express proteins,” Ms. Gualdoni explained.
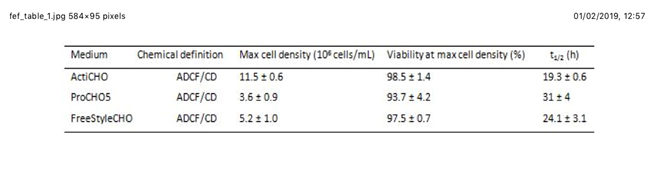
CHO-S growth kinetics in the three commercially available serum-free media tested. Abbreviations: Max: maximum; t1/2: duplication time; ADCF: animal-derived component free; CD: chemically defined; + number: standard deviation of the measured value.
Enhancing upstream productivity
The Novo Nordisk Pharmatech team at Booth 914 at the Santa Clara Convention Center will also showcase feature its recombinant insulin as a key component in growth media for mammalian cells with significant advantages in bioprocess optimization, high cell viability and higher productivity in the upstream manufacturing process.
“These benefits will be highly significant for teams working in upstream bio manufacturing, R&D, and cell line and media development across Asia, looking for bioprocess optimization: high cell viability and high productivity,” Ms. Gualdoni commented.
Unblocking the CAR-T supply chain
The team will also be showing how using high quality recombinant Human Insulin as part of serum replacement can improve T Cell expansion and in-vivo function in CAR-T therapy optimization.
“Transfer of T Cells genetically modified to target cancer cell antigens (CAR-T therapy) is a promising oncology treatment strategy. The big challenge remains to scale-up the manufacturing process, improve consistency and reduce the costs,” said Ms. Gualdoni.
“One of the main current limiting factors in the manufacturing process is dependency on human serum, which is used to support T lymphocyte growth and survival when cultured ex-vivo,” she said.
“Human serum is expensive and the current supply chain will not meet the demands when different approved CAR-T therapies become blockbuster treatments. High focus on T-Cell media development has recently allowed several serum free media being introduced on the market, with different proprietary formulation based on key components, such as human insulin,” said Ms. Gualdoni.
“This is a particularly relevant development for people working in upstream bio manufacturing, R&D, BD and any stakeholder involved in cell line and media development, cell and gene therapy manufacturing who are focused on bioprocess optimization, high cell viability and high productivity,” she concluded.
About Novo Nordisk Pharmatech A/S
Novo Nordisk Pharmatech A/S was established in 1949 as FeF Chemicals, acquired by Novo Nordisk in 1986 and has been part of the pharmaceutical group since then. Novo Nordisk Pharmatech A/S is the leading worldwide supplier of recombinant insulin for cell growth media and pharmaceutical grade quaternary ammonium compounds (quats) for the pharmaceutical, biopharmaceutical industry.
About BioProcess International US West 2019
The BioProcess International US West (BPI West) is one of an international series of BioProcess events that aim to provide leading phase-based bioprocessing knowledge sharing focused on advancing promising biologic developments towards commercial success.
BPI West 3019 is a four-day event opening March 11 at the Santa Clara Convention Center in California.
The meeting will feature e full program of sessions with presentations from leading scientists, engineers and regulators working across the entire bioprocessing spectrum, including Dr. Jerry Murry, Vice President of Amgen, Dr. Rahul Singhvi, Chief Operating Officer of Takeda Vaccines, Inc., Dr. Jens Vogel, President & CEO of Boehringer Ingelheim Fremont Inc. and Dr. Arifa Khan, Supervisory Microbiologist at the US Food & Drug Administration.
The conference is constructed around six scientific tracks covering Viral Safety, Early Stage Process Development, Late Stage PD, Commercial Manufacturing and Single-Use/Flexible Facilities.
The event is organized by KNECT 365 with further information at: https://lifesciences.knect365.com/bpi-west/.
















