By Novo Nordisk…
Novo Nordisk Pharmatech showing recombinant insulin advances at Annual Biologics Manufacturing Asia
Koege, Denmark: – Novo Nordisk Pharmatech A/S, the leading worldwide supplier of high-quality insulin, will be a leading presence at the upcoming Annual Biologics Manufacturing Asia meeting in Singapore.
Novo Nordisk Pharmatech will be an exhibitor at the event with a stand at Booth 8 at the Suntec Singapore Exhibition Center showing latest developments in its powerful recombinant insulin human animal-free (AF) for cell culture, particularly its potential to increase viable Chinese hamster ovary (CHO) cell viability and productivity.
Serum-free demand
“CHO cells are one of the most widely used platforms for the production of biopharmaceuticals. Increased demand for safety and reliability has moved the standard for CHO cell culture media from serum to serum-free and further on to chemically defined media,” commented Novo Nordisk Recombinant Insulin Product Manager, Sara Gualdoni, who will be present in Singapore.
The team in Singapore will also be showcasing Novo Nordisk Pharmatech recombinant insulin as a key component in growth media for mammalian cells with significant advantages in bioprocess optimization, high cell viability and higher productivity in the upstream manufacturing process.
Increased cell density
“In collaboration with UAB, we have shown that adding animal origin free insulin to leading commercially available off-the-shelf chemically defined media has resulted in significant increases in viable cell density,” Ms. Gualdoni explained.
“In addition to this benefit, recombinant insulin has been proven to aid in the expression of difficult to express proteins,” she added.
“These benefits will be highly significant for teams working in upstream bio manufacturing, R&D, and cell line and media development across Asia, looking for bioprocess optimization: high cell viability and high productivity,” Ms. Gualdoni commented.
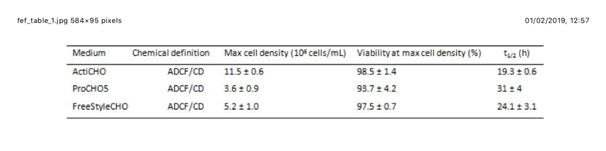
CHO-S growth kinetics in the three commercially available serum-free media tested. Abbreviations: Max: maximum; t1/2: duplication time; ADCF: animal-derived component free; CD: chemically defined; + number: standard deviation of the measured value
About Novo Nordisk Pharmatech A/S
Novo Nordisk Pharmatech A/S was established in 1949 as FeF Chemicals, acquired by Novo Nordisk in 1986 and has been part of the pharmaceutical group since then. Novo Nordisk Pharmatech A/S is the leading worldwide supplier of recombinant insulin for cell growth media and pharmaceutical grade quaternary ammonium compounds (quats) for the pharmaceutical, biopharmaceutical industry.
About Biologics Manufacturing Asia 2019
Biologics Manufacturing Asia bills itself as the only biomanufacturing focused knowledge-sharing platform for the Asia-Pacific region, covering Singapore, South Korea, Taiwan, Japan, Australia, India, Indonesia, Malaysia, and China.
The annual event brings together more than 400 biomanufacturing stakeholders from pharma, biopharma, contract manufacturing organisations, academic & research institutes and regulatory authorities.
The 6th Biologics Manufacturing Asia meeting is a three-day event, opening February 26 at the , Suntec International Convention & Exhibition Center in Singapore.
The event is organized by IMAPAC, with more information at: www.imapac.com/business_conference/biomanufacturing-world-asia-2019/home/.
















