By CBDepot s.r.o.
CBDepot sister company CB21 Pharma launches new cannabidiol-based suppositories to OTC market
Prague, Czech Republic: – CB21 Pharma, sister company of pioneering producer and B2B supplier of upmarket cannabinoid solutions, CBDepot, is launching vaginal and rectal cannabidiol (CBD) suppositories for over-the-counter (OTC) sales in Europe.
The products are certified by the certification body 3EC International and registered as Class IIa medical devices by the Czech State Institute for Drug Control (SUKL.eu).
GMP compliant
The suppositories are produced in CB21 Pharma’s EU GMP compliant manufacturing site, following the system of quality according to EN ISO 13485:2016, rendering them the only fully legal and compliant OTC products containing CBD on the European market.
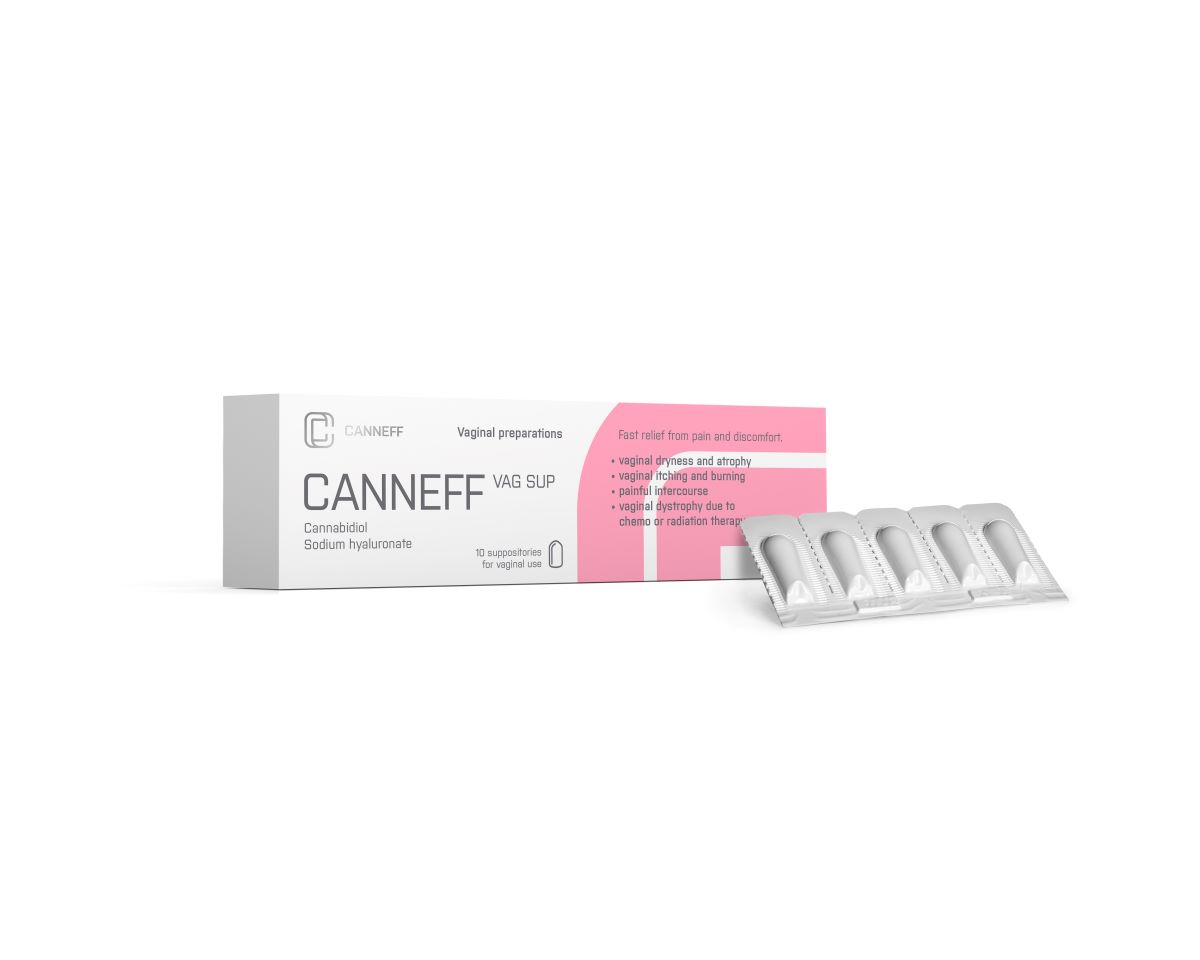
CANNEFF VAG SUP vaginal suppositories are designed to promote relief from the symptoms of vaginal discomfort after treated vaginal inflammation or vaginal mycosis and to heal atrophic and dystrophic vaginal mucous membrane conditions. These can occur after childbirth and gynecological surgery or in dystrophy treatments after chemotherapy, ionizing radiation, or in vaginal dryness due to estrogen deficiency.
CANNEFF SUP rectal suppositories are used as a supplemental treatment in the anorectal canal conditions’ healing processes after proctological operations; internal and external hemorrhoids; proctitis; cryptitis; anal fissures; perianal fistula.
Distribution opportunities
“We encourage distributors of medical devices and OTC products from the EU member states to get in touch with us and seize the opportunity to bring these unique products to their domestic markets,“ said CB21 Pharma CEO Jan Storch.
“CB21 Pharma is currently designing clinical studies with the CANNEFF suppositories for medical conditions of autoimmune diseases, aiming for the registration of the products as prescription medical products,” adds Jan Storch.
About CBDepot
CBDepot is an innovative and recognized producer and B2B supplier of upmarket natural and synthesized cannabinoid ingredients to fast-growing markets in the food, cosmetics, pharmaceuticals and health products industries sectors.
CBDepot specializes in bulk supply of cannabinoid-based ingredients for food, cosmetics and pharmaceutical sectors. Its products ensure that content of tetrahydrocannabinol (THC), the main intoxicating element of cannabis, is below detectable levels. Instead, its products emphasize the two other main natural ingredients, cannabidiol (CBD) and cannabigerol (CBG).
Established in 2014, CBDepot has become a leader in helping to establish legal and quality standards for cannabinoid ingredients and the formulation of cannabidiol derivatives. Since foundation it has operated under legal license granted by the government of the Czech Republic.
The company’s initial mission was the supply of “Legal and Quality CBD” at a time when sourcing ethical and quality isolated CBD was a challenge. CBDepot’s scientific knowledge is based on close links with the Czech Academy of Sciences.
The company’s Chief Sales Officer, Boris Baňas, is an ex-board member of the European Industrial Hemp Association (EIHA), where he successfully campaigned for the establishment of new Europe-wide standards.
About CB21 Pharma
Prague-based CB21 Pharma s.r.o. (CB21) specializes in the research and development of pharmaceutical grade cannabinoid-based products, dedicated to bringing CANNEFF-branded products to the market in line with the evidence-based medicinal concepts.
CB21 CANNEFF products include cosmetic lines and medical devices that combine CBD with other active ingredients to achieve synergistic benefits.
CB21 is a full service cannabis company committed to excellence from extraction and fractionation of the plant material to high-tech purification of cannabinoids.
All CB21 Pharma products are manufactured in accordance with the correctly implemented Pharmaceutical Quality System incorporating GMP standards and Quality Risk Management.
Production is in collaboration with the international quality control company EUROFINS BEL/NOVAMANN s.r.o, whose laboratories are fully authorized by State Institute for Drug Control Bratislava, Slovak Republic for pharmaceutical testing.
Find out more at: https://cb21pharma.com/en/