By TAmiRNA GmbH
TAmiRNA offers SARS CoV-2 antibody kit to aid in Covid-19 immunity testing
Biotech innovator TAmiRNA is distributing a lateral flow antibody test kit to detect IgM and IgG antibodies against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) as a valuable aid to immunity testing for Covid-19.
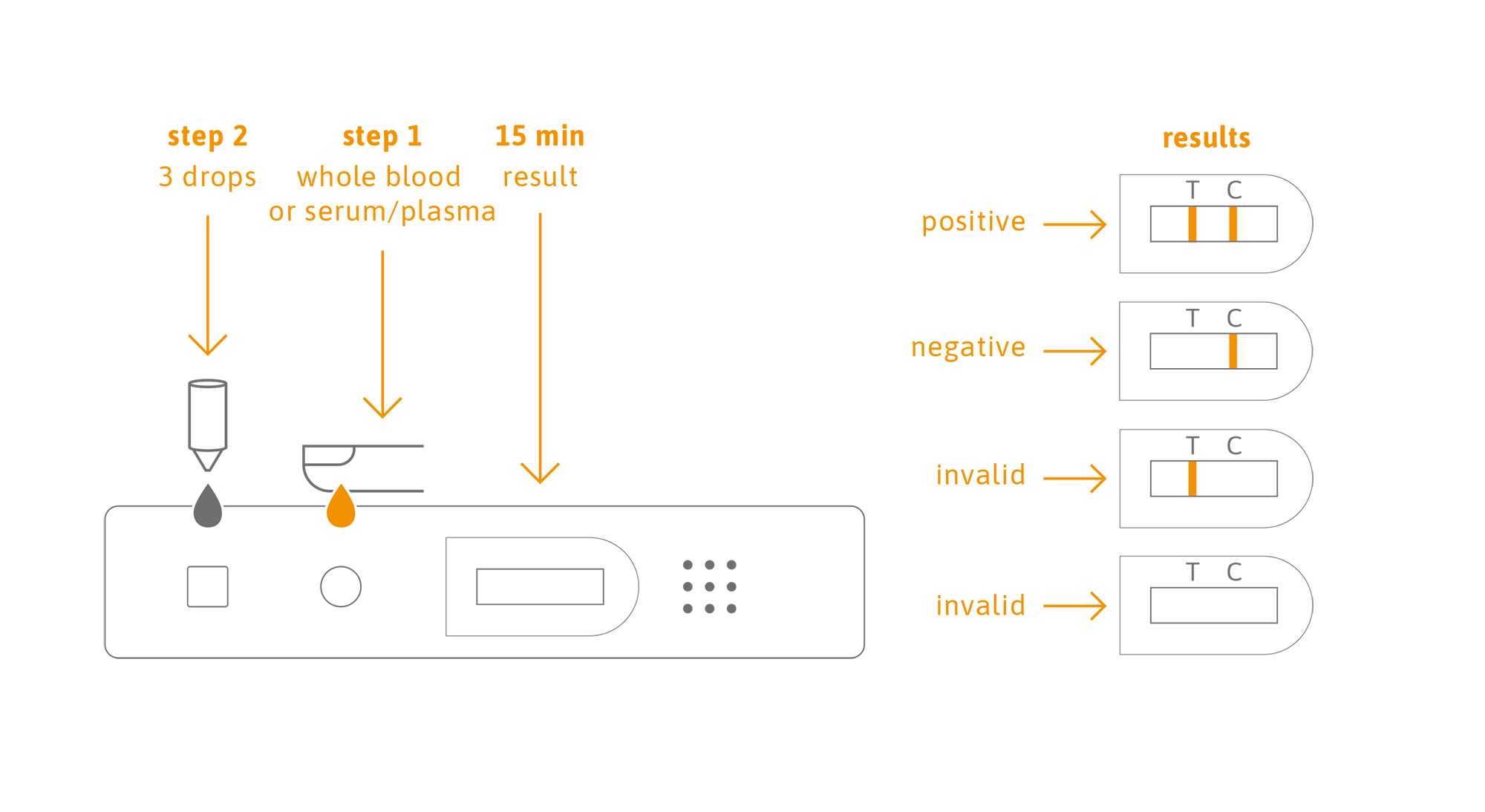
The fast-acting test allows results to be returned within 15 minutes and can be used as an aid in identification of those people who have developed antibody response to coronavirus infection disease 2019 (COVID-19), which is caused by SARS-CoV-2.
Fast-acting CoV-2 immunochromatographic assay
The SARS-CoV-2 Antibody Lateral Flow Test is an immunochromatographic assay for rapid, qualitative detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) IgM & IgG antibodies in human whole capillary or venous blood, serum or plasma sample.
The assay is intended for research use only by hospitals, academic research groups, clinical research groups, pharmaceutical companies and laboratories. It provides information about an ongoing or overcome infection with SARS-CoV-2, but cannot be used as the sole basis for treatment or other management decision.
CoV-2 antibody test features
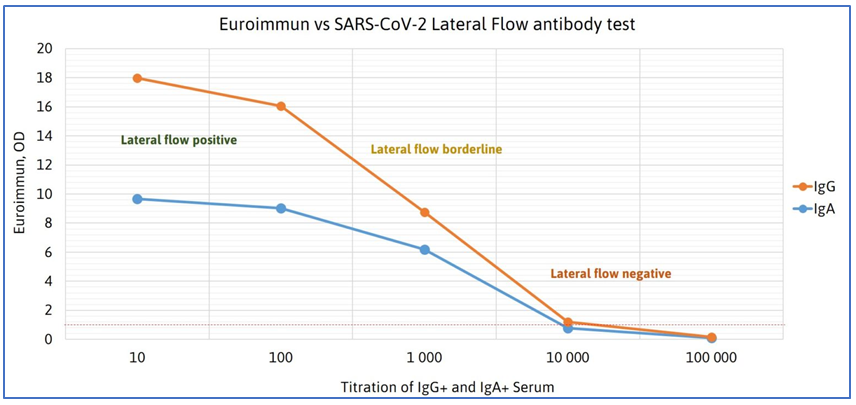
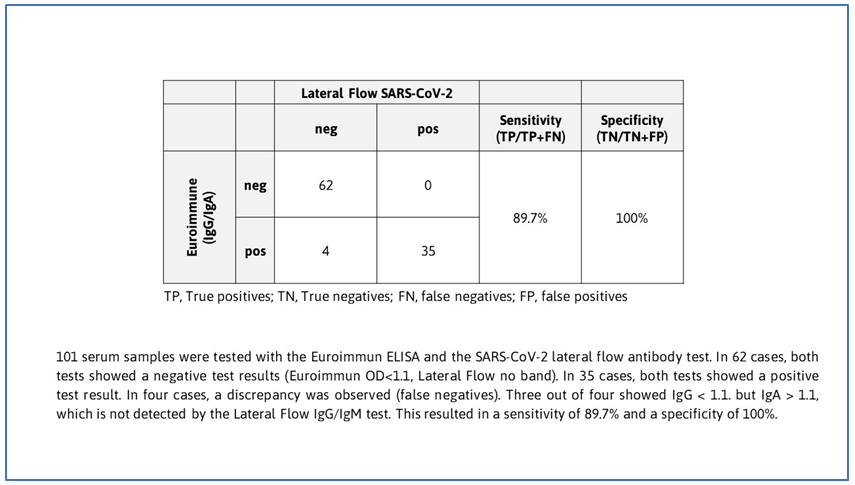
SARS-CoV-2 Antibody Test (Lateral Flow Method) is intended for rapid (15 minutes) detection of IgM or IgG antibodies against SARS-CoV-2. The test sensitivity and specificity were recently evaluated in cooperation with the “Agentur für Gesundheit und Ernährungssicherheit GmbH (AGES)”. Based on a total of 101 serum samples a sensitivity of 89.7% and a specificity of 100% were observed.
The assay requires:
- 10 μL of serum and plasma
- 20 μL of venous or capillary whole blood (fingerstick or plasma tubes)
This kit is based on the principle of capture immunoassay for determination of SARS-CoV-2 IgG/IgM antibodies in human whole blood (venous and capillary), serum and plasma. When the specimen is added into the test device, the specimen is absorbed into the device by capillary action, mixes with the SARS-CoV-2 antigen-dye conjugate and flows across the pre-coated membrane.
When the SARS-CoV-2 antibody level in the specimen are at or above the target cutoff (test detection limit), the antibodies bound to the antigen-dye conjugate are captured by anti-human IgG antibody and anti-human μ chain antibody immobilized in the Test Region (T) of the device, and this produces a colored test band that indicates a positive result. When the SARS-CoV-2 antibody level in the specimen is zero or below the target cutoff, there is not a visible colored band in the Test Region (T) of the device. This indicates a negative result.
To serve as a procedure control, a colored line will appear at the Control Region (C), if the test has been performed properly.
About TAmiRNA
TAmiRNA specializes in technologies for profiling levels of blood-circulating microRNAs and developing multi-parametric classification algorithms (“signatures”). TAmiRNA uses these technologies to develop minimal-invasive diagnostic tests for drug development, early diagnosis and prognosis of disease, and as companion diagnostic tests to support treatment decisions.
More information available at: www.tamirna.com
Resources
Click on SARS-CoV-2 Antibody Lateral Flow Test for technical details.
Click on TAmiRNA CoV-2 testing service for further details