By PHC Europe…
PHC Europe BV
Netherlands-based PHC Europe BV (PHCEU) is the European and African arm of the international PHC Corporation, whose PHCbi products are recognized worldwide as a leading and trusted brand for reliable, high-quality medical and laboratory equipment, with particular focus on technologies for preservation, incubation, sterilization and healthcare.
The company’s mission is to provide a leading, trusted brand for sustainable healthcare and biomedical product solutions, supporting the work of its customers to improve the health and well-being of people around the world.
PHCEU solutions range from excellent sample security in an ultra-low temperature (ULT) freezer or the best environment for cell culture in a CO2 incubator, while its relationships span the pharmaceutical, healthcare and biotechnology sectors, with a pan-continental supply and support network that can quickly meet customer demands across Europe and Africa.
Services
PHCbi products are grouped in four main ranges: Preservation, Incubation, Processing and Sterilization.
Preservation
The Preservation Business Segment encompasses six main product lines:
- Ultra-Low Freezers: PHC offers a complete range of freezers that operate between -70°C and -86°C. These incorporate advanced technologies, such as the TwinGuard dual cooling system for ultimate sample security and ecologically-friendly natural hydrocarbon (HC) refrigerants for lower energy consumption, reduced environmental impact and substantially lower running costs. PHCbi ULT freezers boast industry-leading reputations for performance and reliability as well as the highest levels of service and technical support.
- Cryogenic ULT freezers: operating at -152°C for long term storage of sensitive, high value samples. PHCbi was a pioneer in cryopreservation solutions from the early 1990s and has extended its leadership with improved insulation, better refrigeration technology and alarm systems and increased capacity. The latest Cryogenic MDF ultra-low temperature freezers offer unparalleled safety, reliability and uniform temperatures.
- Biomedical Freezers: Operating between -30°C and -40°C, these freezers are suited for storage of vaccines, enzymes and diagnostic samples, as well as temperature and aging tests.
- Pharmaceutical Refrigerators that offer uniform storage temperature for demanding pharmaceutical, medicinal, vaccine, and other temperature-sensitive applications.
- Blood Bank Refrigerators that conform to internationally recognized AABB (formerly American Association of Blood Banks) criteria, with assured stable and reliable temperature control utilizing proprietary PHCbi technology, with high efficiency compressors delivering more rapid cooling and quieter performance.
- Liquid Nitrogen Cryopreservation, including Cryosystem and Isothermal liquid nitrogen freezers, lab tanks, transport containers and vapor shippers.
Incubation
The five principal product ranges in the PHCbi incubation segment comprise:
- CO2 Incubators: Designed for incubating cell cultures with precision, security and ease of use and incorporating innovative technologies such as built-in UV lamp, InCu-saFe® design to eliminate contamination sources, Direct Heat and Air Jacket system and Dual IR sensor to regulate temperature and CO2 levels.
- Multigas Incubators: designed for a broad range of biomedical, pharmaceutical, medical and clinical research applications with more precise control of O2 and CO2 levels, along with patented Direct Heat and Air Jacket Heating Systems, to promote stable cell culture environment.
- Heated Incubators: providing precise and stable incubation environments between +5°C and +80°C for a wide range of biological research, environmental study and other applications.
- Cooled Incubators: programmable cooled Incubators offering exceptional accuracy, reliability and flexibility meeting test and experimentation protocols requiring wide-ranging, high-precision, automatic temperature and lighting controls.
- Climatic Test Chambers: suitable for a wide range of research, environmental studies and testing applications requiring programmable temperature, humidity and lighting patterns.
Cell Culture Plates
- 3D Plates and Dishes
PHCEU provides superior quality three-dimensional cell culture platforms with a variety of well shapes to enable spheroid culturing of your specific cell type. PrimeSurface® cell culture labware are ultra-low attachment (ULA) dishes and plates that promote scaffold free, self-assembly of spheroid formation. The plates are pre-coated with unique ultra-hydrophilic polymer that enables spontaneous spheroid formation of uniform size and shape. The ULA plates have high optical clarity making them highly suitable for bright field imaging and confocal microscopy. In addition to the widely used 96 well U bottom plate, 96 well plates are also available in V and M bottom, giving scientists a choice to form tighter spheroids that are needed for specific cell types. For high throughput screening (HTS) needs, 384 well plates are available in clear and white. - Slit well plate
PHCEU is offering a new slit-well, ultra-low attachment 3D plate to facilitate easy handling of media exchange without disrupting spheroid formation. Cell culturing involves frequent media replacement to provide nutrition to growing cells. In a standard 96 well ultra low cell attachment plate, media aspiration or dispensing has to be done extremely carefully to avoid disturbing the unattached spheroid, making this a time consuming operation. With the introduction of PrimeSurface 96 Slit-Well Plate, media exchange for 96 well plates can be efficiently handled with one step dispensing or aspiration for all 96 wells. This product can decrease pipetting time by over 80% while minimizing the risk of spheroid damage.
Sterilization
PHCbi Sterilization offers a range of portable laboratory autoclaves in various capacities designed for compact profile, easy operation and maximum safety. They provide a safe and reliable high-pressure steam sterilizing environment with microprocessor control to ensure correct temperature is accurately and easily maintained. Customizable sterilization programs can adjust temperature and time with multiple safety systems to protect against overpressure, overheating or door errors.
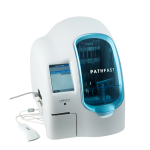
PATHFAST™
PATHFAST™ system combines the accuracy of a full-scale lab analyser with the flexibility of a mobile solution. Its compact design and low weight makes PATHFAST™ the ideal analysis system in laboratories, hospitals and medical offices available wherever fast quantitative results are required at the point of care. With its space-saving design and large degree of flexibility PATHFAST™ is also an ideal supplement or back-up system in central labs. It can be available at any time (24 hrs per day) without interfering with the processes of routine analysis. PATHFAST™ is providing fast, differential diagnosis and is easy to operate, install and network. High precision makes this analyser an adequate “satellite” of a full-scale lab on a cardiology, intensive care or emergency ward. Parallel processing enables the examination of six samples in only a few minutes from whole blood, serum or plasma.
Structure and history
PHCEU is part of the international PHC Corporation, founded in Japan in 1969, that now operates across more than 120 countries worldwide, focusing on the three core business fields of Medical Devices, Healthcare IT & Life Sciences.
PHCEU was founded in 1990 as the European sales arm of PHC, headquartered at Den Haag in the Netherlands with principal operations located at Etten-Leur in The Netherlands, Avon in France and Loughborough in the UK. The company employs some 85 people across Europe.
Currently PHCEU has direct sales operations with dedicated customer service teams in the Netherlands, France and UK, with sales forces and distributors in Germany, Switzerland and Italy, soon to be expanded to three further countries. In total, it has more than 50 distributors in 32 countries, including Russia, Israel and Turkey and is expanding its reach across EMEA are already therefor Algeria, Morocco, Tunisia, Ivory Coast, South Africa, Uganda, Kenya, Botswana, Zambia, Tanzania, Mauritius, Ethiopia and Egypt.
Quality and Regulatory
PHC products and facilities are certified to ISO 9001, ISO 14001 and ISO 13485 standards for quality, environmental management and medical devices.
PHC can provide turnkey solutions for validation and qualification in accordance with all current GMPs, GLPs, GCPs, 21 CFR Part 11, PAT, ISO and specific customer requirements and applications.
Because key component parts are designed and built by PHC, it can offer precise and in-depth validation resources specific to PHCbi laboratory products. PHCEU can also provide comprehensive expertise in laboratory equipment to exact compliance needs. PHCEU validation systems employ advanced technology coupled with the latest trends to insure compliance with accurate and time efficient completion.