By Evercyte GmbH
Evercyte GmbH
Austria-based Evercyte GmbH manufacture a range of cell-based products, including telomerized human cells. While characterized by an unlimited growth potential, Evercyte cell cultures maintain a phenotype similar to the corresponding primary cells. Therefore, Evercyte cells show an optimal balance between growth and differentiation, which is of ever-increasing importance for various applications in biotechnology and biomedicine.
Within the product portfolio of Evercyte there are different cell types (from epithelial cells to mesenchymal stem cells) isolated from a large variety of tissues and organs. Moreover, Evercyte developed urine as non-invasive, universal cell source for the isolation of stem cells as well as for the establishment of patient-derived induced pluripotent stem cells or telomerized kidney podocytes. Extracellular vesicles from mesenchymal stem cells (e.g. from bone-marrow, Wharton´s Jelly, adipose tissue, chorionic plate, amniotic membrane, endometrium) or any other telomerized cell line are available upon request.
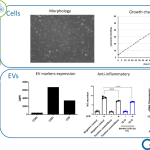
With this optimal balance between growth and differentiation Evercyte cells can be used to set-up relevant and standardizable in vitro test systems with applications in various fields of biomedicine, toxicology and biotechnology with the aim to help its clients’ reduce time and money spent on drug discovery and development.
Due to their unique characteristics, Evercyte cells also represent cell factories for manufacturing of complex molecules, whereby a focus has been put on production of extracellular vesicles / exosomes that are currently evaluated for their therapeutic potential in tissue regeneration and as drug targeting vehicle.
As contract research organization (CRO), Evercyte offers services to develop customer-tailored cell lines, bioassay development as well as testing drug candidates for efficacy, potency, toxicity as well as a potential senolytic / senomorphic activities. Production and characterization of extracellular vesicles from customer-derived cell culture are also offered to our customers.
Evercyte offers its human cells to academic and/or non-profit-organizations as well as to companies (profit-organizations) by granting licenses for the use of Evercyte material.
Academic or non-profit-organization clients
Evercyte grants a license for a single cell line not for human use, for research purpose only, for non-commercial use in client’s laboratory only for indefinite time (please ask for the license fee of a specific cell line or register on www.evercyte.com).
Profit-organization clients
Evercyte grants licenses for an initial six-month period, for research purpose only to for profit-organization. The initial license period of six months shall be followed by an annual license period, which renews automatically each year.
Evercyte also grants licenses for the use of mesenchymal stem cells that qualify for production of clinical grade extracellular vesicles for human application.