By Bioassay GmbH
Bioassay GmbH
Bioassay GmbH is a bioanalytical contract laboratory (CRO) specializing in the development, validation and routine performance of bioanalytical assays in the areas of Pharmacology and Toxicology.
Bioassay serves clients in the biotechnology, chemistry and cosmetics sectors and is particularly expert in carrying out potency assays under GMP for biopharmaceuticals, including therapeutic antibodies, hormones and cytokines.
Activities/Products
Bioassay offers a range of services based on in-vivo and in-vitro systems including potency assays (GMP) and toxicity Studies (GLP).
The company has developed a range of proprietary customized, product specific, assays in accordance to ICH and FDA guidelines.
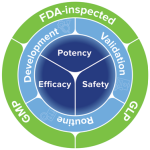
The Bioassay services portfolio focuses on four key areas:
- Biopharma Potency Bioassay GmbH executes and assesses potency assays for quality control of active pharmaceutical ingredients and drugs. It also possesses extensive experience in developing and validating new assays under ICH-Guidelines, as well as optimizing existing assay formats for transfer into specific applications. Bioassay GmbH has established a series of cGMP potency assays for antibodies, Rituximab, Trastuzumab, Adalimumab and Infliximab.
- Biopharma Efficacy Bioassay GmbH has established efficacy models to address the demand for characterization of new drugs, especially in biopharmaceutical fields like monoclonal antibodies, hormones and cytokines. Bioassay also has extended experience in characterizing biosimilars and modified biologics, such as pegylated products, and provides specialized models for characterizing nephrology and diabetes drug candidates.Cell-based Analysis services include FACS binding assays, specific cell line development and neutralizing antibodies assays. Bioassay has also developed a portfolio of Pharmacokinetic & Disease Models that include pharmacokinetic & metabolic studies and specific Nephrology & Diabetic Models, including:
-
- Acute Kidney Injury (AKI) / Ischemia-Reperfusion-Injury (IRI)
- Kidney transplantation model
- 5/6 Nephrectomy (Chronic Kidney Disease)
- Hypertension Model (e.g. renal artery stenosis)
- Unilateral Ureteral Obstruction (UUO)
- Diabetic nephropathy
- Biopharma Safety
Bioassay offers a panel of assays for assessment of complex biotherapeutics ranging from monoclonal antibodies to recombinant proteins to characterize their interaction with different molecules in the human body to filter out possibilities of unwanted effects that might prejudice safety profile or compromise therapeutic efficacy. Biologics safety panels cover antibody-dependent cellular cytotoxicity (ADCC), neutralizing antibodies, Multi-Cytokine Analysis, Immuntoxicology (ITOX3), skin sensitization (LLNA, h-Clat & MUSST) and toxicokinetics. - Chemicals / Cosmetics Safety Bioassay GmbH runs acute cGLP toxicity and skin sensitization studies for the chemicals and cosmetic industries. It was selected by the European Commission Joint Research Centre to participate in a project whose aim is to deliver validated alternative methods and strategies for skin sensitisation testing to be used by chemical and cosmetic industries for the safety assessment of products. We worked in close contact with multinational companies as well as the European Commission Joint Research Centre for the initial assessment of two of those tests involving flow-cytometry methods. The company provides standardized assay methods for chemical and cosmetic products safety in acute toxicity, including dermal toxicity (OECD 402), dermal Irritation (OECD 404), eye Irritation (OECD 405), oral toxicity (OECD 423), toxicity prediction (BALB/c 3T3 Neutral Red Uptake Cytotoxicity Test).It also offers skin sensitization assays for chemicals (OECD 442B), cosmetics (H-Clat Assay), cosmetics (MUSST Assay), and chemicals (OECD 406).
Structure and Background
Bioassay GmbH was privately founded in 2002 as a team of five people primarily focused on performing an Erythropoietin bioassay. Bioassay was acquired by Reaction Biology Corporation in 2022, and continues to support its portfolio of clients and remains committed to its standard of excellence.
Initially located at the BASF facilities in Ludwigshafen, Bioassay moved to its current location at Heidelberg’s Technology Park in 2005, where it now forms part of a cluster of innovative biotech companies and institutions with links to Heidelberg University. The Bioassay workforce now exceeds 20 people.
The laboratory was cGMP certified in 2002, cGLP certified in 2007 and passed successive FDA inspections in 2012 and 2015.
The company’s principal shareholders, Dr. Sven-Michael Cords and Dr. Axel Hohenstein, are also its managing directors. Dr. Cords joined the company in 2006 to lead in-vivo studies and acts as Chief Operating Officer, while Dr. Hohenstein joined the following year as head of in-vitro studies. He is also Bioassay’s Chief Scientific Officer.
Quality, Regulatory and Standards
Bioassay carries out and documents all procedures in compliance with GLP and GMP standards, which also form the basis for its quality standards, guaranteeing acceptability of the data generated in its laboratories.
Regular Inspection of quality standards carried out by regulatory authorities and in customer audits encourage continuous improvement and reinforce transparency of our performance.
Main processes for ensuring quality include SOPs, internal Quality Manual written to GMP, GLP and OECD guidelines. Bioassay also works to ICH and US Food & Drug Administration standards, holding FDA registration following inspections.
Bioassay operates from advanced 1000 square meter in vitro and in vivo laboratories, with all devices IQ, OQ and PQ qualified and analytical methods validated in accordance to ICH guidelines.